 
Calculate the entropy change for 1. This example problem demonstrates how to examine the reactants and products to predict the sign of the change in entropy of a reaction. 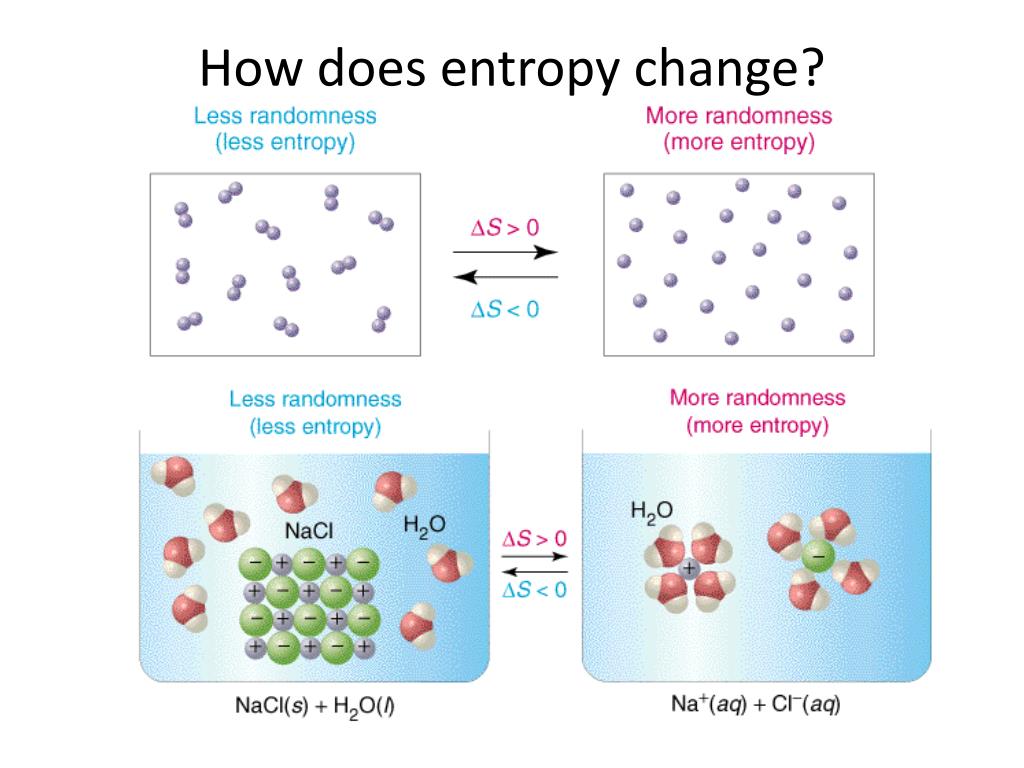
It is easy to lose a sign during thermochemistry homework problems. The enthalpy of fusion for water is 6.01 kJ/mol. For problems involving changes in entropy, knowing if the change should be positive or negative is a useful tool to check your work. Although this result was obtained for a particular case, its validity can be shown to be far more general: There is no net change in the entropy of a system undergoing any complete reversible cyclic process. The entropy change for a phase change at constant pressure is given by. What is the entropy change during the expansion of a van der Waals gas for which CV is a constant The equation of state for one mole of. The entropy change for this reaction is highly negative because three gaseous molecules are being converted into two liquid. There is no net change in the entropy of the Carnot engine over a complete cycle. The entropy change for the formation of liquid water from gaseous hydrogen and oxygen can be calculated using this equation: 2H 2(g) + O 2(g) 2H 2O(l) So 2(69.9) 2(131.0) + 1(205.0) 327 J/K mol. 
Figure 19.3.2: A Generalized Plot of Entropy versus Temperature for a Single Substance. However, we know that for a Carnot engine, Unlike enthalpy or internal energy, it is possible to obtain absolute entropy values by measuring the entropy change that occurs between the reference point of 0 K corresponding to S 0 J/ (molK) and 298 K. The equation for the change of entropy (delta S) of a system or object is the energy transferred to or from the object (Q), measured in Joules, divided by the average temperature of the object (T. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
